SEASPINE

Atoll™ OCT System
Seamless Transition from Occiput to Sacrum
The Atoll™ OCT System exemplifies the company’s commitment to their longstanding design philosophy; “Fewer Parts, Fewer Steps”. The Atoll™ OCT System contains minimal instruments, eight of which are multifunctional.
The Atoll™ OCT System is comprised of polyaxial screws, hooks, rods, locking screw assemblies, and connectors. The system components may be rigidly locked together in a variety of configurations to promote fusion for a wide variety of patient anatomies.
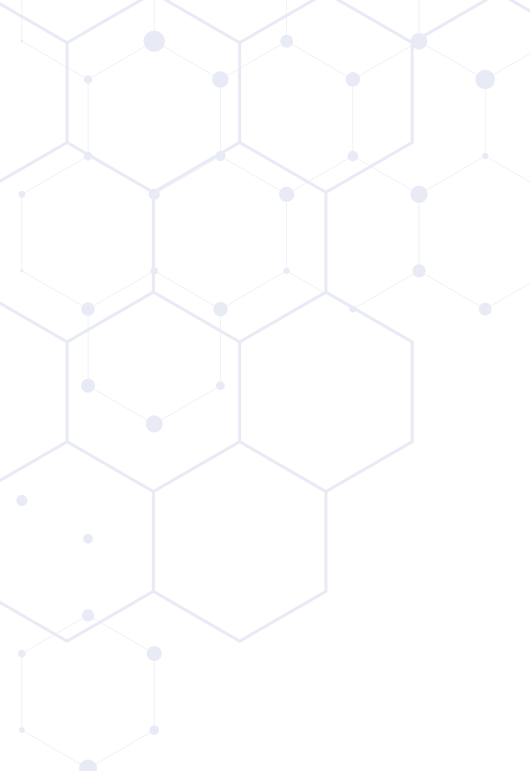
Occiptal Set
Plate allows for up to 5 points of fixation (3 midline and 2 lateral)
OCT Spinal System
More intra-operative options to meet the patient’s anatomical needs and surgical technique preferences
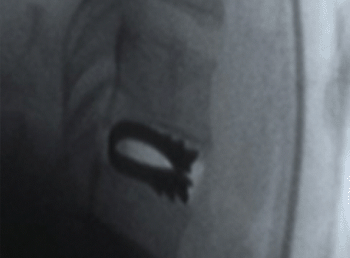
Transition Set
Allows easy connection to the thoracolumbar region with the Coral Spinal System.

Vu L•POD™ (IBD Lumbar Systems)
The Vu L•POD™ Intervertebral Body Fusion Device (IBD) is designed to deliver a system that provides the optimum graft area with a wide array of implants and instruments.
The Vu L•POD™’s highly engineered 3D convex surfaces closely match the anatomy of a vertebral endplate

BodyForm™ (Lumbar Lateral Plating System)
Thoracic Fixation System in conjunction with BodyForm™ Thoraco-Lumbar Fixation System provides a fixation solution for spine regions from T1-L5.
The BodyFormTM Thoraco-Lumbar and BodyFormTM Thoracic Fixation Systems consist of a variety of shapes and sizes of plates, screw lengths, and setscrews. There are also ancillary instrument sets. All implant components are machined from implant grade titanium alloy, Ti-6Al-4V (ELI) per ASTM F-136.
PARADIGM SPINE


Coflex®
The first and only Motion Preserving Minimally Invasive Treatment Approved for Moderate to Severe Spinal Stenosis After Decompression
The coflex® Interlaminar Technology is an Interlaminar Stabilization™ device indicated for use in one or two level lumbar stenosis from L1-L5 in skeletally mature patients with at least moderate impairment in function, who experience relief in flexion from their symptoms of leg/buttocks/groin pain, with or without back pain, and who have undergone at least 6 months of non-operative treatment. The coflex® is intended to be implanted midline between adjacent lamina of 1 or 2 contiguous lumbar motion segments.
The Interlaminar Stabilization™ device is implanted after decompression of stenosis at the affected level(s).
Anatomical Design :
- Excellent endplate accommodation for primary stability.
- Atraumatic anchorage to avoid heterotopic ossification.
- Teeth located anteriorly for secure anchorage.
- Three implant heights for appropriate height restoration.
- Four different footprints for proper endplate coverage.
Single-Piece Implant:
- Excellent fatigue strength.
- No wear debris.
- Titanium alloy.
Ease of Use:
- Standard surgical technique.
- Easy instrumentation.
- Implant insertion under compression (reduced height).
- Depth stop for trial implants and insertion instrument.
Axial Compliance:
- Shock absorption capabilities.
- Compressible in flexion.
Functionally Dynamic:
- Physiological center of rotation.
- Controlled rotational stability.


Coflex F®
Designed to Deliver Surgeon Confidence and Patient Satisfaction
The coflex-F stabilization system is a posterior, non-pedicle supplemental fixation device intended for use with an interbody cage as an adjunct to fusion at a single level in the lumbar spine (LI – SI). It is intended for attachment to the spinous processes for the purpose of achieving stabilization to promote fusion in patients with degenerative disc disease – defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies with up to Grade I spondylolisthesis.
The minimally invasive system is designed to allow for a low-profile, tissue-friendly, anatomical fit.
Ease of Use:
- Simple surgical technique.
- Intuitive instrumentation.
Reduced Surgical Risks:
- Excellent safety profile.
- Protection of neural structures.
Reduced Cost:
- Shorter operating time.
- Faster patient rehabilitation.
Reduced Iatrogenic Trauma:
- Less muscle trauma and blood loss.
- Smaller skin incision.
Design Features:
- Interlaminar positioning.
- Secure anchorage through rivet fixation.
- Large contact area for optimized stress distribution.
- Five anatomical sizes.
- Color-coded instrumentation.
- Titanium alloy that is biocompatible.
DSS®
Customized Stabilization for a Variety of Spinal Pathologies
The DSS® Stabilization System is intended as a single-level system for non-cervical pedicle fixation from the T4 to S1 vertebrae to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion in the treatment of acute and chronic instabilities or deformities of the thoracic, lumbar, and sacral spine. The system consists of polyaxial, cannulated pedicle screws; slotted couplers; and rigid couplers.
The DSS® System allows various fixation configurations to address patients with an array of lumbar spine anatomies and pathologies. Each construct is designed to treat specific indications and has its own limitations. It is important to review the cleared Instructions for Use for a complete list of indications, warnings, precautions, contraindications, and limitations for the rigid and slotted coupler designs.




HPS®
Lowers Stresses and Reduces Risk of Adjacent Segment Disease
HPS™ is designed to combine modern, rigid spinal fixation with a topping-off option. It results in less stress on adjacent segments and a reduced risk of developing adjacent segment disease, which causes new symptoms after spine surgery. The key element, the DSS® coupler, is the most efficient coupler available on the market and has been clinically proven in about 5,000 cases all around the world.
The combination of the coupler with tulip screws and rods makes the Hybrid Performance System the most versatile and universal system available on the market, allowing for mono- and multi-level fusions. The low-profile, high-quality polyaxial screws and ergonomic instruments enable fast and optimal implant positioning and deformity correction techniques.
CENTINEL SPINE
Stalif-Midline® (Lumbar)
No-Profile® Anterior Lumbar Integrated Interbody System
STALIF MIDLINE® is part of an “integrated” system of implants and instruments designed to create consistent clinical results and avoid more destructive multiple incision surgeries.
Designed to simplify surgery and reduce the number of surgical steps required.
No Supplemental Fixation
Integrated cage and screw system is cleared for use without supplemental fixation. Some surgeons, however, may still choose to use plates or pedicle screws.
Conforms to Patient Anatomy
The “No-Profile” design allows it to nest fully within the confines of the vertebral body, leaving the anatomy unchanged external to the interbody.
Compressive Fixation Ensures Graft and Endplate Contact As the preferred design to ensure fixation in cancellous vertebral bone, cancellous screws—combined with our screw head design—produce a lag effect between the vertebral body and the implanted device.
The lag effect provides constant compressive forces against the implant, which results in a greater interface between the graft and the endplate to facilitate the fusion process.
Engineered to Maximize Opportunities for Fusion The 3-screw design utilizes a midline convergence pattern in conjunction with an optimal horizontal inclination. These integrated elements conform to AO principles of fracture fixation, increasing the possibility of a fusion2.




Stalif-C® (Cervical)
STALIF C® is part of an “integrated” system of implants and instruments designed to create consistent clinical results and avoid more destructive multiple incision surgeries.
Efficient Design
Fewer, Simpler Surgical Steps
STALIF C® was designed to simplify surgery and reduce the number of surgical steps required.
No Supplemental Fixation
STALIF C’s integrated cage and screw system is cleared for use without supplemental fixation. Some surgeons, however, may still choose to use anterior plates or posterior fixation.
No-Profile®.
Conforms to Patient Anatomy
The STALIF C® “No-Profile” design allows it to nest fully within the confines of the vertebral body, leaving the anatomy unchanged external to the interbody.
The Lag Effect.
Compressive Fixation Ensures Graft and Endplate Contact
As the preferred design to ensure fixation in cancellous vertebral bone, cancellous screws combined with our screw head design produce a lag effect between the vertebral body and the implanted device.
The lag effect provides constant compressive forces against the implant which results in a greater interface between the graft and the endplate to facilitate the fusion process.
Optimal Screw Trajectories.
Engineered to Maximize Opportunities for Fusion
The STALIF C® 3-screw design utilizes a midline convergence pattern in conjunction with an optimal horizontal inclination. These integrated elements conform to AO principles of fracture fixation, increasing the possibility of a fusion2
TECRES
Mendec Spine®
High Viscosity- All in one mixer
The first all in one mixing system preloaded with new High Viscosity spine resin. Mendec Spine HV System is a new closed mixer that already has both powder and liquid components inside. Tecres keeps on its history of firsts presenting a new milestone in spine market. Mendec Spine HV System proudly follows the original patent of Cemex System, the first bone cement all in one system.


Resin
Application Kit
Mendec Spine Kit
Specific resin for vertebral consolidation + device for injection A specific high fluidity resin with high x-ray contrast; A device which allows the application into vertebral bodies in a safe and accurate manner.

TEDAN SURGICAL INNOVATION
Surgical Instrumentation
Tedan Surgical Innovations, LLC, is dedicated to providing innovative, high quality specialty surgical instrumentation for use in orthopedic, neuro and thoracic surgery. TSI guarantees that all of our products have been manufactured by skilled instrument craftsmen, using only quality materials. Every effort is made to manufacture the finest quality surgical instruments at exacting specifications.
TeDan Surgical Innovations specializes in cervical retractors, lumbar retractors, thoracic retractors, anterior hip retractors, distraction screws, minimally invasive retractors and instruments for minimally invasive surgery.
CARE FUSION (V. MUELLER®)

Chroma-Line® Kerrison Rongeurs
Our unique rongeurs feature:
Titanium nitride coating that reduces friction between metal parts for smooth and effective cutting color coded insert that easily identifies the bite size.
Chroma-Line® Spinal Curettes
Have been designed with the surgeon’s concern in mind. Ergonomically designed handles provide maximum control. Deep cup design color coded handle system reduces cup size selection time, allowing for efficient and timely completion of surgical procedures full range of cup sizes from 6-0 to 6 is available with straight, angle, reverse-angle and 90 angle shafts.